Health
FDA Memo Reveals Link Between Child Deaths and COVID-19 Vaccines
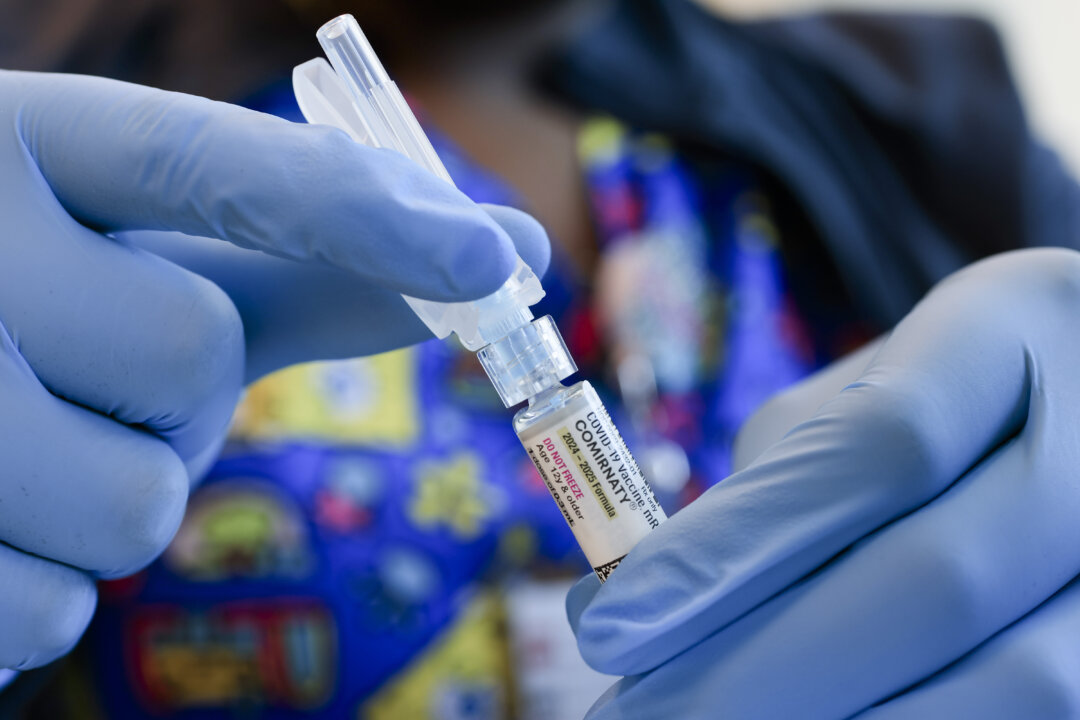
An internal memo from the Food and Drug Administration (FDA) indicates a troubling connection between COVID-19 vaccinations and child fatalities. According to the document, obtained by The Epoch Times, scientists within the agency’s vaccine-safety offices have identified “no fewer than 10” child deaths that they believe are related to the vaccine. This revelation marks a significant acknowledgment within the FDA regarding potential risks associated with COVID-19 vaccinations.
The memo was authored by Dr. Vinay Prasad, director of the FDA’s Center for Biologics Evaluation and Research (CBER). It highlights findings from a review conducted by the Office of Biostatistics and Pharmacovigilance, examining reports submitted to the Vaccine Adverse Event Reporting System (VAERS) between 2021 and 2024. The investigation focused on 96 death reports, with the staff concluding that at least 10 of these cases met the criteria for a likely, probable, or possible causal link to COVID-19 vaccination.
Review Findings and Implications
The internal review sheds light on the potential adverse effects of vaccinations, particularly among children. The FDA memo suggests that the findings are part of an ongoing evaluation of vaccine safety, aimed at ensuring that the benefits of vaccination outweigh any associated risks.
The deaths identified in the memo are part of a broader discussion about vaccine safety, especially as public health officials continue to advocate for COVID-19 vaccinations among children. The acknowledgment from FDA officials highlights concerns that have been expressed by some medical professionals and parents regarding the safety of vaccines for younger populations.
Dr. Prasad’s memo has sparked discussions about transparency and the importance of monitoring vaccine-related adverse events. The findings have drawn attention to the need for ongoing research and communication regarding the potential risks of vaccinations, particularly in vulnerable populations.
Context and Future Considerations
The FDA has been at the forefront of approving and regulating vaccines during the pandemic, emphasizing their effectiveness in preventing severe illness and death. However, the emergence of internal documents suggesting potential links to fatalities raises critical questions about safety protocols and monitoring systems in place.
Public health authorities are tasked with balancing the urgency of vaccination campaigns against the necessity of ensuring safety and efficacy. As more data becomes available, it will be vital to continue evaluating the benefits and risks associated with COVID-19 vaccinations, especially for children.
The discussions prompted by this memo underscore the importance of transparency in public health communications. Parents and guardians must be well-informed about the potential risks and benefits, allowing them to make educated decisions regarding vaccinations for their children.
As the situation evolves, the FDA’s response and further investigations will likely shape public perception and policy surrounding COVID-19 vaccinations in pediatric populations.
-

 Science4 weeks ago
Science4 weeks agoUniversity of Hawaiʻi Joins $25.6M AI Project to Monitor Disasters
-

 Business1 month ago
Business1 month agoForeign Inflows into Japan Stocks Surge to ¥1.34 Trillion
-

 Top Stories1 month ago
Top Stories1 month agoBOYNEXTDOOR’s Jaehyun Faces Backlash Amid BTS-TWICE Controversy
-

 Top Stories1 month ago
Top Stories1 month agoCarson Wentz Out for Season After Shoulder Surgery: Urgent Update
-

 Top Stories1 month ago
Top Stories1 month agoMarc Buoniconti’s Legacy: 40 Years Later, Lives Transformed
-

 Health1 month ago
Health1 month agoInnovative Surgery Restores Confidence for Breast Cancer Patients
-

 Sports2 months ago
Sports2 months agoSteve Kerr Supports Jonathan Kuminga After Ejection in Preseason Game
-

 Lifestyle2 months ago
Lifestyle2 months agoKelsea Ballerini Launches ‘Burn the Baggage’ Candle with Ranger Station
-

 Science2 months ago
Science2 months agoChicago’s Viral ‘Rat Hole’ Likely Created by Squirrel, Study Reveals
-

 Entertainment2 months ago
Entertainment2 months agoZoe Saldana Advocates for James Cameron’s Avatar Documentary
-

 Lifestyle2 months ago
Lifestyle2 months agoDua Lipa Celebrates Passing GCSE Spanish During World Tour
-

 Business2 months ago
Business2 months agoTyler Technologies Set to Reveal Q3 2025 Earnings on October 22









